|
Olimab 60 Mg Injection
Olimab 60 Mg Injection Specification
- Storage Instructions
- Store in Refrigerator (2C - 8C), Do Not Freeze
- Features
- Ready to Use, High Purity, Pre-filled Syringe
- Frequency
- As Directed by Physician (typically once every 6 months)
- Function
- Anti-resorptive, RANKL Inhibitor
- Accuracy
- Pharmaceutical Grade Dosage Accuracy
- Shelf Life
- 36 Months
- Instruments Type
- Pre-filled Syringe
- Usage Type
- Hospital, Clinical, Prescription Only
- Measurement Range
- 60 mg/1 mL
- Equipment Type
- Pharmaceutical Injection
- Material
- Sterile Liquid
- Condition
- New
- Technology
- Biotechnology Derived Monoclonal Antibody
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Parenteral (Injection)
- Use
- Treatment of Osteoporosis
- Dimension (L*W*H)
- Single Dose Pre-filled Syringe
- Weight
- Prefilled Syringe Packaging (approx. few grams)
- Color
- Clear to Slightly Yellow Solution
- Brand Name
- Olimab 60 Mg Injection
- Marketed Strength
- 60 mg Denosumab in 1 mL solution
- Packaging Type
- Single Use Prefilled Syringe
- MRP (As per packaging)
- As per local regulation
- Product Code
- Olimab 60
- Indications
- Postmenopausal Osteoporosis, Increased Risk of Fractures
- Prescription Required
- Yes
- Inactive Ingredients
- Sorbitol, Acetate, Polysorbate 20, Water for injection
- Route of Administration
- Subcutaneous Injection
- Active Ingredient
- Denosumab 60 mg
- Dosage Form
- Injection
Olimab 60 Mg Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Olimab 60 Mg Injection
Olimab 60 mg injection contains omalizumab as its active ingredient, which is a monoclonal antibody used to treat certain allergic conditions, particularly moderate to severe asthma and chronic urticaria (hives) that are not well controlled with other medications. Omalizumab works by binding to immunoglobulin E (IgE), an antibody involved in allergic reactions, thereby preventing it from interacting with its receptors on immune cells. This helps to reduce inflammation and allergic responses in the body.
For asthma, Olimab is typically prescribed to patients whose asthma is not controlled with inhaled corticosteroids or other asthma medications. It can help reduce the frequency of asthma attacks, improve lung function, and reduce the need for oral steroids. In chronic urticaria, it helps reduce the severity and frequency of hives and itching in patients who do not respond to antihistamine therapy.
The injection is usually administered once every 2 or 4 weeks, depending on the specific condition being treated and the patient's response to the medication. It is given as a subcutaneous (under the skin) injection by a healthcare provider, and the exact dose is determined based on factors such as the patients weight and the severity of the condition being treated.
Like any medication, Olimab 60 mg injection may cause side effects. Common side effects include reactions at the injection site (such as redness, swelling, or pain), headache, and upper respiratory infections. Less commonly, it may cause more serious side effects such as an allergic reaction, which can include symptoms like difficulty breathing, swelling of the face or throat, or rash. It is important to contact a healthcare provider immediately if any of these symptoms occur.
Patients should inform their doctor about any pre-existing conditions, such as a history of anaphylaxis, before starting treatment with Olimab. It is also important to continue using other prescribed asthma medications or allergy treatments unless otherwise directed by the healthcare provider.
Precision Therapy for Osteoporosis
Olimab 60 Mg Injection is a pharmaceutical-grade treatment developed for women with postmenopausal osteoporosis, especially those at heightened risk of fractures. Using advanced biotechnology, each pre-filled syringe delivers a precise 60 mg dose of denosumab, targeting bone loss via RANKL inhibition. Rigorous purity and ready-to-use packaging maximize patient safety and convenience.
Convenient and Controlled Administration
With its pre-filled syringe form, Olimab 60 Mg Injection ensures ease and accuracy. Administered subcutaneously by trained healthcare providers, this format minimizes preparation time and risk of dosing errors. The clear to slightly yellow sterile liquid is stable for up to 36 months when stored at the recommended refrigeration temperature, offering reliability in clinical settings.
FAQ's of Olimab 60 Mg Injection:
Q: How does Olimab 60 Mg Injection work in treating osteoporosis?
A: Olimab 60 Mg Injection contains denosumab, a monoclonal antibody that inhibits RANKL-a protein involved in bone resorption. By blocking this protein, the medicine helps to reduce bone loss, increase bone density, and lower the risk of fractures in postmenopausal women.Q: What is the recommended frequency for using Olimab 60 Mg Injection?
A: Olimab 60 Mg Injection is typically administered once every six months as directed by your physician. The exact schedule may vary depending on individual patient needs and medical advice.Q: Where should Olimab 60 Mg Injection be administered?
A: Olimab 60 Mg Injection is designed for subcutaneous injection and should be administered by a qualified healthcare professional, usually in a hospital or clinical environment.Q: What are the main benefits of Olimab 60 Mg Injection?
A: The main benefits include a reduction in bone loss, an increase in bone strength, and a significantly decreased risk of fractures in individuals with postmenopausal osteoporosis. Its pre-filled, single-use syringe ensures precise dosing and easy administration.Q: How should Olimab 60 Mg Injection be stored?
A: Olimab 60 Mg Injection must be kept refrigerated at 2C to 8C and must not be frozen. The pre-filled syringe should be stored in its original packaging to protect it from light until use.Q: What should I do if a dose of Olimab 60 Mg Injection is missed?
A: If you miss a scheduled dose, contact your healthcare provider as soon as possible. The next dose should be administered as recommended by your doctor to maintain treatment efficacy.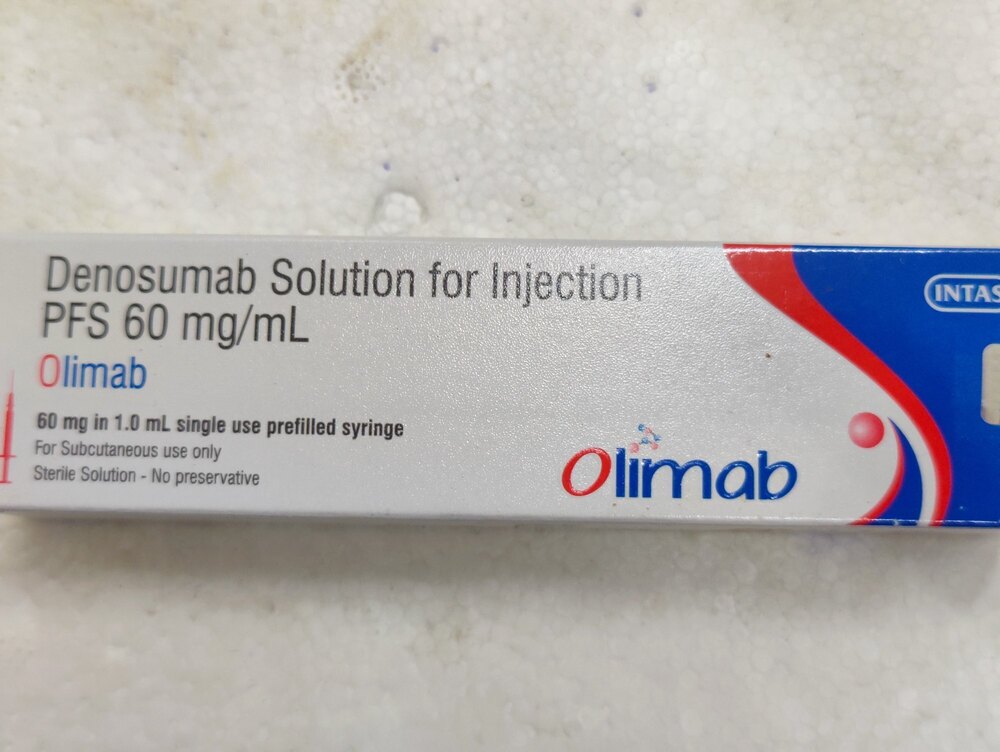

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
Magic Touch Sirolimus Coated Balloon Catheter
Price 6400 INR / Pack
Minimum Order Quantity : 100 Packs
Use : Treatment of coronary artery disease and instent restenosis
Operating Type : Other, Manual/Interventional
Technology : Sirolimus Drug Coating using Nanolute Technology
Wall Mounted : No
Translumina Introducer Needle 20g
Price 870 INR / Pack
Minimum Order Quantity : 100 Packs
Use : Vascular Access, Catheter Introduction
Operating Type : Manual
Technology : Precision Bevel Needle Technology
Wall Mounted : No
Dr. Surgical Y Push-Pull non-removable
Price 550 INR / Pack
Minimum Order Quantity : 100 Packs
Use : Surgical Applications
Operating Type : Manual
Technology : Manual Mechanical
Wall Mounted : No
Coloplast Alterna Free Convex Deep base Plate 17761
Price 300 INR / Piece
Minimum Order Quantity : 100 Pieces
Use : Colostomy / Ileostomy / Urostomy Care
Operating Type : Other, Manual Application
Technology : TwoPiece System
Wall Mounted : No
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |