|
Mircera Injection 75mg
Mircera Injection 75mg Specification
- Shelf Life
- 24 Months
- Storage Instructions
- Store at 2C to 8C (Refrigerated), Do not freeze
- Measurement Range
- 75mg (per syringe/vial)
- Usage Type
- Medical/Clinical
- Instruments Type
- Injection
- Frequency
- As directed by physician (usually monthly or as required)
- Features
- Long-acting Erythropoiesis-Stimulating Agent
- Accuracy
- Standardized Pharmaceutical Dosage
- Function
- Stimulates erythropoiesis (Red Blood Cell Production)
- Equipment Type
- Pharmaceutical Injection
- Material
- Sterile Liquid Solution
- Condition
- New
- Technology
- Biotechnology (Recombinant DNA Technology)
- Portable
- Yes (Single-use Pre-filled Syringe)
- Wall Mounted
- No
- Operating Type
- Parenteral/Injectable
- Use
- Treatment of Anemia associated with Chronic Kidney Disease (CKD)
- Dimension (L*W*H)
- Pre-filled Syringe/Injection Vial
- Color
- Clear Colorless Solution
- Administration Route
- Subcutaneous or Intravenous
- Disposal Instructions
- Dispose of used syringes safely in sharps container
- Expiry Date
- Stamped on packaging
- ATC Code
- B03XA03
- Active Ingredient
- Methoxy Polyethylene Glycol-Epoetin Beta
- Caution
- Use under medical supervision only
- Compatibility
- Compatible with standard subcutaneous and intravenous administration protocols
- Prescription Required
- Yes
- Batch Number
- Mentioned on packaging
- Indication
- Anemia treatment in CKD including dialysis and non-dialysis patients
- Packaging Size
- 75mg/0.3mL single dose pre-filled syringe
Mircera Injection 75mg Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Mircera Injection 75mg
Mircera (75 mg injection) is a brand name for methoxy polyethylene glycol-epoetin beta, a medication used primarily to treat anemia associated with chronic kidney disease (CKD) and other conditions where red blood cell production is impaired.
Mircera is a long-acting erythropoiesis-stimulating agent (ESA) that stimulates the bone marrow to produce more red blood cells. It works by mimicking the action of erythropoietin, a naturally occurring hormone that stimulates red blood cell production in response to low oxygen levels in the body.
Key uses of Mircera:
- Chronic kidney disease (CKD) related anemia: It is used to treat anemia in people with chronic kidney disease, especially for those on dialysis or not on dialysis.
- Cancer-related anemia: It may be prescribed to treat anemia caused by chemotherapy.
How it works:
Mircera is injected into the bloodstream, typically once a month or as prescribed by your doctor. The long-acting formulation means fewer injections compared to other erythropoiesis-stimulating agents.
Common side effects of Mircera:
- High blood pressure (due to increased red blood cell production)
- Headaches
- Joint or muscle pain
- Fatigue
- Nausea
- Injection site reactions (pain, redness, or irritation)
- Increased risk of blood clots (especially in patients with underlying cardiovascular conditions)
Important considerations:
- Monitoring: Your doctor will monitor your hemoglobin levels, iron levels, and blood pressure while you are on Mircera to ensure the treatment is working effectively and safely.
- Iron supplementation: Since erythropoiesis-stimulating agents like Mircera require iron to make new red blood cells, your doctor may recommend iron supplements or iron-rich foods to prevent iron deficiency.
As with any medication, it's important to follow the prescribed dosing regimen and inform your healthcare provider of any side effects or new health conditions during treatment.
Advanced Treatment for CKD-Related Anemia
Mircera Injection 75mg is designed specifically to address anemia in patients with chronic kidney disease, whether on dialysis or not. As a long-acting agent, it reduces the frequency of injections needed compared to traditional therapies. By stimulating red blood cell production, Mircera helps improve energy levels, reduce fatigue, and enhance overall wellbeing.
Safe and Convenient Administration
Mircera comes in a pre-filled, sterile syringe for ease of use, making it portable and convenient for both clinic-based and home administration. Administration-either subcutaneously or intravenously-should always be performed by or under the supervision of a healthcare professional to ensure dosing accuracy and patient safety.
Proper Storage and Handling
To preserve its effectiveness, Mircera should be stored in a refrigerator at 2C to 8C, away from freezing temperatures. Always check the expiry date and batch number on the packaging before use. Used syringes should be discarded safely in an approved sharps container to prevent accidental injury or contamination.
FAQ's of Mircera Injection 75mg:
Q: How does Mircera Injection 75mg help treat anemia in chronic kidney disease?
A: Mircera contains Methoxy Polyethylene Glycol-Epoetin Beta, which stimulates the bone marrow to produce more red blood cells, thereby correcting anemia commonly found in chronic kidney disease (CKD) patients.Q: What is the recommended frequency and route of administering Mircera?
A: Mircera is typically administered once monthly or as directed by your physician. It can be given as a subcutaneous (under the skin) or intravenous (into a vein) injection by a healthcare professional.Q: Where should I store Mircera pre-filled syringes before use?
A: Mircera should be stored in a refrigerator at a temperature between 2C and 8C. Do not freeze the solution, and always keep it in its original packaging until use.Q: What safety precautions should be followed when disposing of used Mircera syringes?
A: Used pre-filled syringes must be disposed of in a designated sharps container to minimize the risk of needlestick injuries or contamination. Follow your healthcare provider's disposal instructions closely.Q: When should Mircera not be used?
A: Mircera should never be used past its expiry date, which is indicated on the packaging. Also, it must only be used under the supervision of a qualified healthcare professional and as prescribed.Q: What are the main benefits of using Mircera over other anemia treatments?
A: Mircera is a long-acting erythropoiesis stimulant, requiring fewer injections compared to other treatments. This reduces hospital visits, improves convenience, and helps maintain stable hemoglobin levels in patients with CKD.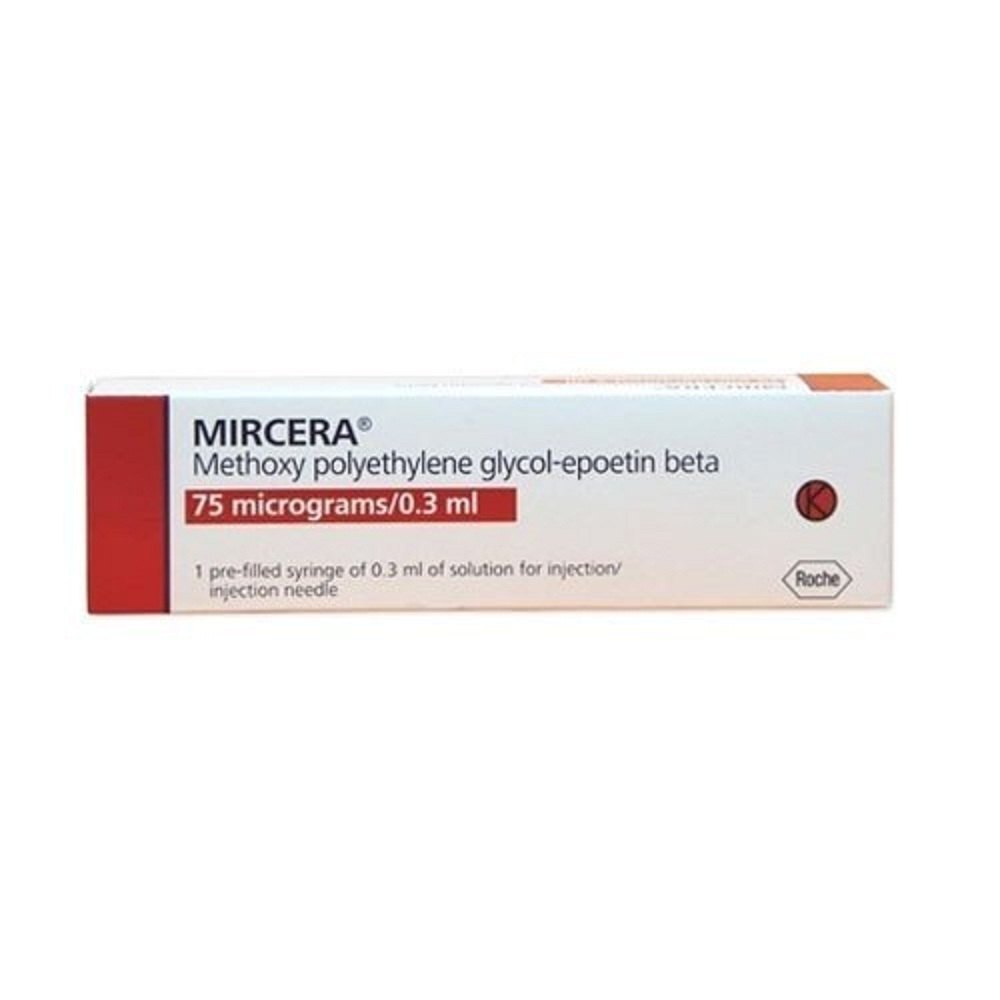

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
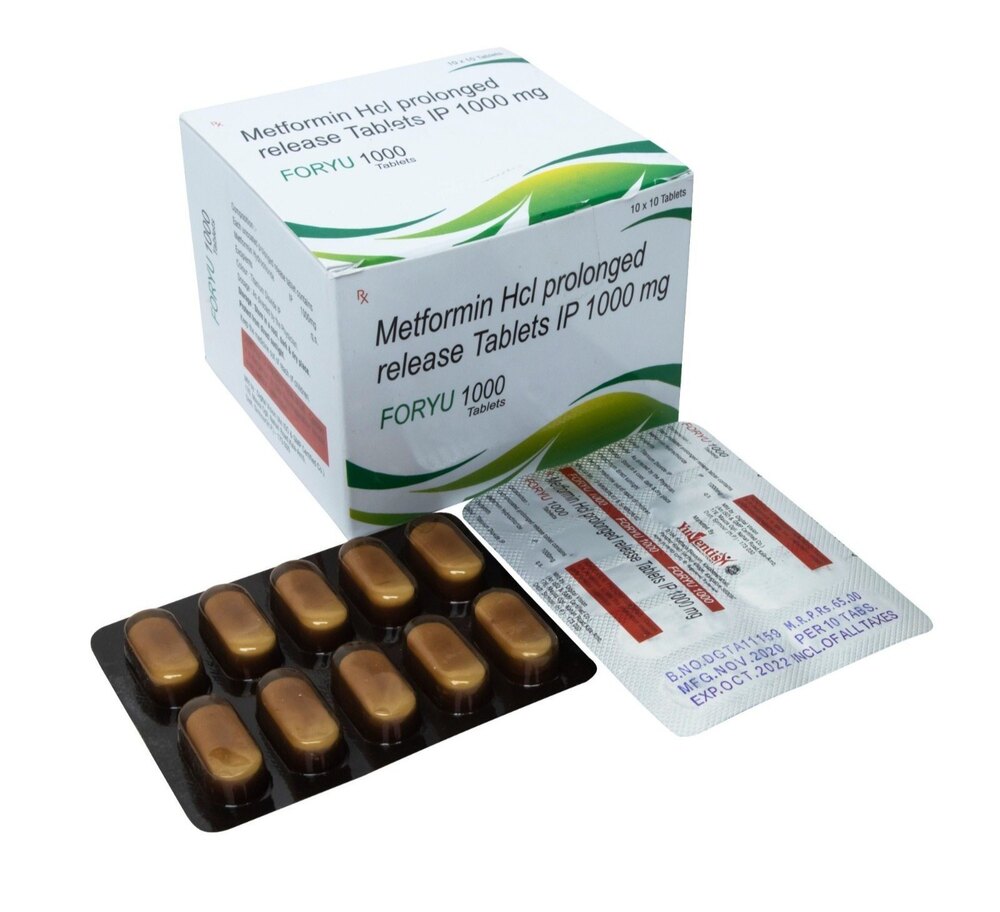
Metformin Tablets BP 1000mg
Price 300 INR / Pack
Minimum Order Quantity : 100 Packs
Operating Type : Other, Oral Administration
Color : White
Shelf Life : 36 months from manufacturing date
Portable : Yes
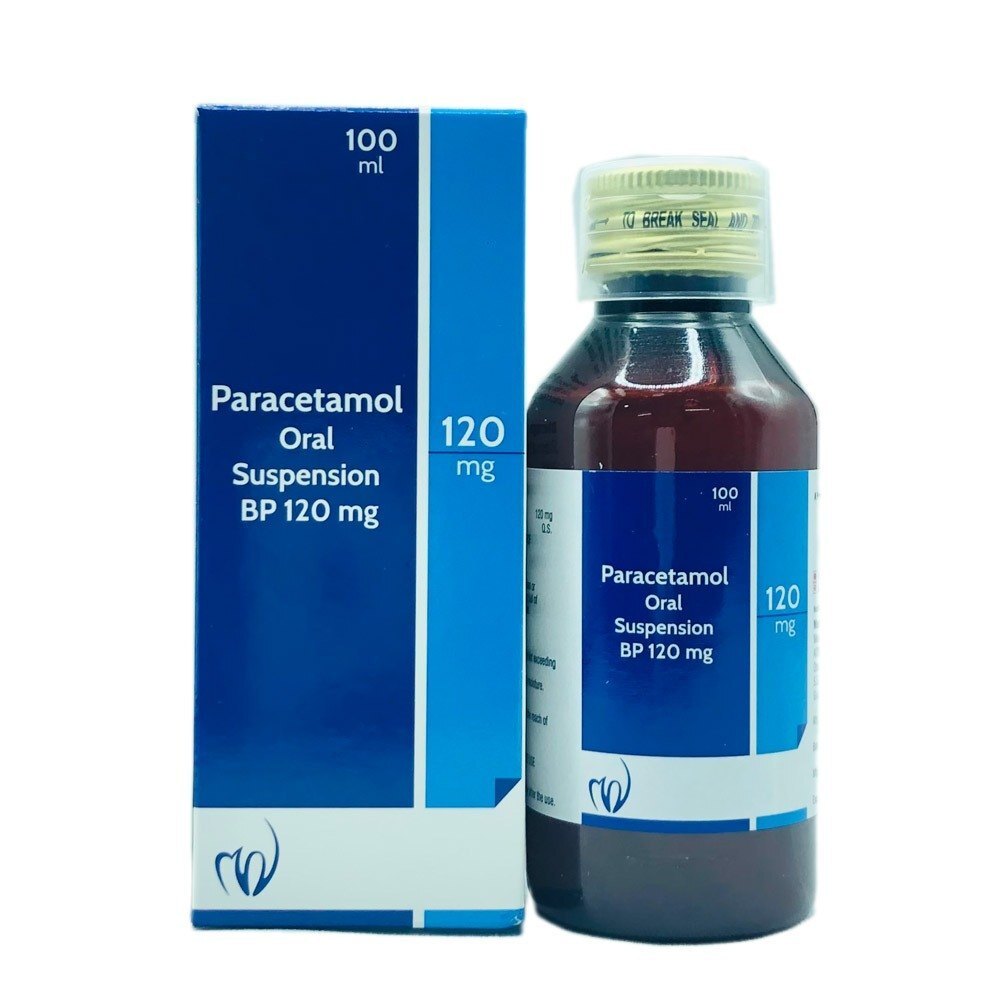
Paracetamol Oral Suspension BP 120mg/5ml
Price 50 INR / Pack
Minimum Order Quantity : 100 Packs
Operating Type : Other, Oral Use
Color : Pink (as per image)
Shelf Life : 24 Months
Portable : Yes
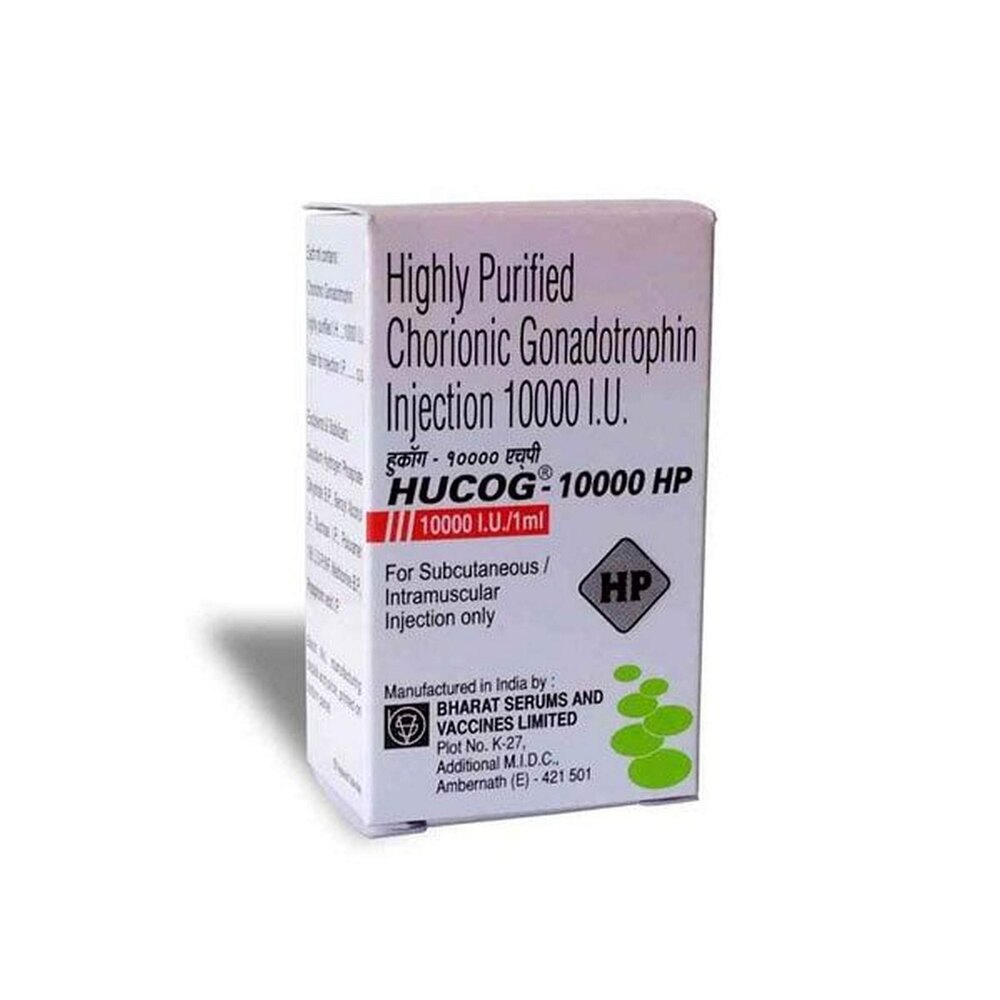
HUCOG 10K INJECTION
Price 5000 INR / Pack
Minimum Order Quantity : 100 Packs
Operating Type : Other, Parenteral (Intramuscular or Subcutaneous) administration
Color : White lyophilized powder
Shelf Life : As indicated on the carton, typically 2436 months
Portable : Yes
Fluconazole Capsules 150mg
Price 15 INR / Pack
Minimum Order Quantity : 100 Packs
Operating Type : Other, Oral
Color : White/Blue
Shelf Life : 24 Months
Portable : Yes
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |