|
Bortecad 2 Mg Injection
Bortecad 2 Mg Injection Specification
- Accuracy
- Pharmaceutical grade
- Measurement Range
- 2 mg per vial
- Shelf Life
- 24 months
- Instruments Type
- Injectable formulation
- Function
- Anticancer treatment
- Features
- Single-use vial, highly potent, sterile preparation
- Storage Instructions
- Store below 25C, protect from light
- Frequency
- As prescribed by physician
- Usage Type
- Hospital and clinical use
- Equipment Type
- Pharmaceutical Injection
- Material
- Lyophilized powder for solution
- Condition
- New
- Technology
- Cytotoxic Agent - Proteasome Inhibitor
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Operating Type
- Parenteral (Intravenous/Subcutaneous)
- Use
- Treatment of multiple myeloma and mantle cell lymphoma
- Dimension (L*W*H)
- Single vial pack
- Weight
- 2 mg per vial
- Color
- White to off-white powder
- Packaging Type
- Glass vial with flip-off seal in box
- Handling Precautions
- Cytotoxic - Handle with care
- Box Contents
- 1 vial of Bortecad 2 mg
- Prescription Required
- Yes
- Dosage Form
- Lyophilized powder for solution for injection
- ATC Code
- L01XX32
- Route of Administration
- IV or SC
- Diluent Required
- Yes, reconstitute before use
- Manufactured by
- Cadila Healthcare Ltd. (Zydus)
- Marketed By
- Zydus Cadila
- Single/Multidose
- Single-dose vial
- Indication
- Multiple myeloma, Mantle cell lymphoma
- Generic Name
- Bortezomib
Bortecad 2 Mg Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Bortecad 2 Mg Injection
Bortecad 2 mg Injection is a brand name for an injectable form of Bortezomib, the same active ingredient found in Borviz. Like Borviz, Bortezomib is a proteasome inhibitor used in the treatment of certain types of cancer, including multiple myeloma and mantle cell lymphoma.
Here are the key details:
Active Ingredient:
- Bortezomib (2 mg per injection)
Common Uses:
- Multiple Myeloma: A type of cancer affecting plasma cells in the bone marrow.
- Mantle Cell Lymphoma: A form of non-Hodgkin lymphoma, a type of cancer that originates in the lymphatic system.
Mechanism of Action:
- Proteasome Inhibition: Bortezomib works by blocking the proteasome, which is responsible for degrading unneeded or damaged proteins in cells. This inhibition leads to the accumulation of damaged proteins, causing cancer cells to die, as they are unable to cope with the stress.
Dosage:
- The dosage and frequency of Bortezomib injections depend on the patients specific condition, overall health, and treatment response.
- It is often administered via intravenous (IV) injection or subcutaneous (under the skin) injection. The standard dosage and schedule must be determined by the oncologist.
Side Effects:
- Common Side Effects:
- Nausea, vomiting, diarrhea
- Fatigue or weakness
- Low blood counts (anemia, thrombocytopenia, neutropenia)
- Fever
- Serious Side Effects:
- Peripheral neuropathy (nerve damage causing numbness, tingling, or pain, especially in the hands and feet)
- Heart problems, such as heart failure or arrhythmias
- Liver or kidney problems
- Blood clotting issues
Precautions:
- Monitoring: Patients receiving Bortezomib should be closely monitored for side effects, especially related to nerve function and blood cell counts.
- It is important to inform the healthcare provider of any pre-existing conditions, such as heart disease, liver or kidney issues, or previous infections, before starting treatment.
Administration:
- The injection should be administered by a healthcare professional in a clinical setting to monitor for immediate reactions.
Key Features and Usage
Bortecad 2 Mg Injection stands out for its high potency and targeted action against cancer cells. As a single-use, sterile pharmaceutical preparation, it delivers Bortezomib-a proteasome inhibitor-directly into the bloodstream. Used primarily in hospitals and clinics, it is employed for the effective management of multiple myeloma and mantle cell lymphoma. Its accurate dosage form and convenient glass vial packaging ensure reliable clinical outcomes.
Administration and Handling
Bortecad must be handled with special precautions due to its cytotoxic properties. The lyophilized powder requires reconstitution with an appropriate diluent before it can be administered through IV or SC injection. Healthcare professionals are responsible for preparing and delivering the dose according to each patient's prescription. The product is packaged for single-dose use, which minimizes contamination risks and maintains pharmaceutical integrity.
Storage and Safety Precautions
To preserve Bortecad's efficacy, store the vial below 25C and protect it from light. Only qualified personnel should handle or administer the drug, wearing appropriate protective equipment due to its cytotoxic nature. The single-use vial should be discarded properly after use, following standard cytotoxic waste protocols, thereby ensuring patient and staff safety while maintaining regulatory compliance.
FAQ's of Bortecad 2 Mg Injection:
Q: How should Bortecad 2 Mg Injection be prepared and administered?
A: Bortecad 2 Mg Injection must be reconstituted with the recommended diluent by a healthcare professional before administration. Once prepared, it is delivered either intravenously (IV) or subcutaneously (SC) as per the physician's instructions.Q: What conditions is Bortecad 2 Mg Injection used to treat?
A: Bortecad is indicated for the treatment of multiple myeloma and mantle cell lymphoma. Its active ingredient, Bortezomib, works as a proteasome inhibitor to target and destroy cancer cells.Q: When should Bortecad 2 Mg Injection be administered during therapy?
A: The exact timing and frequency of Bortecad administration will be determined by a physician based on the patient's condition and treatment plan. Follow your healthcare provider's instructions closely for optimal results.Q: Where should Bortecad 2 Mg Injection be stored prior to use?
A: Store Bortecad below 25C, away from direct light, in its original packaging until ready for use. Proper storage ensures product stability and efficacy until the expiration date.Q: What precautions must be taken when handling Bortecad 2 Mg Injection?
A: Because Bortecad is a cytotoxic agent, it should be prepared and administered by trained medical personnel using protective equipment and following strict handling and disposal protocols.Q: How does Bortecad 2 Mg Injection benefit patients with cancer?
A: Bortecad works by inhibiting proteasomes, leading to the death of cancerous cells and slowing disease progression. This targeted mechanism can enhance treatment outcomes for eligible patients.Q: Is Bortecad 2 Mg Injection suitable for home administration?
A: No, Bortecad should only be prepared and administered in hospitals or clinical settings by healthcare professionals due to its potency and the need for special handling.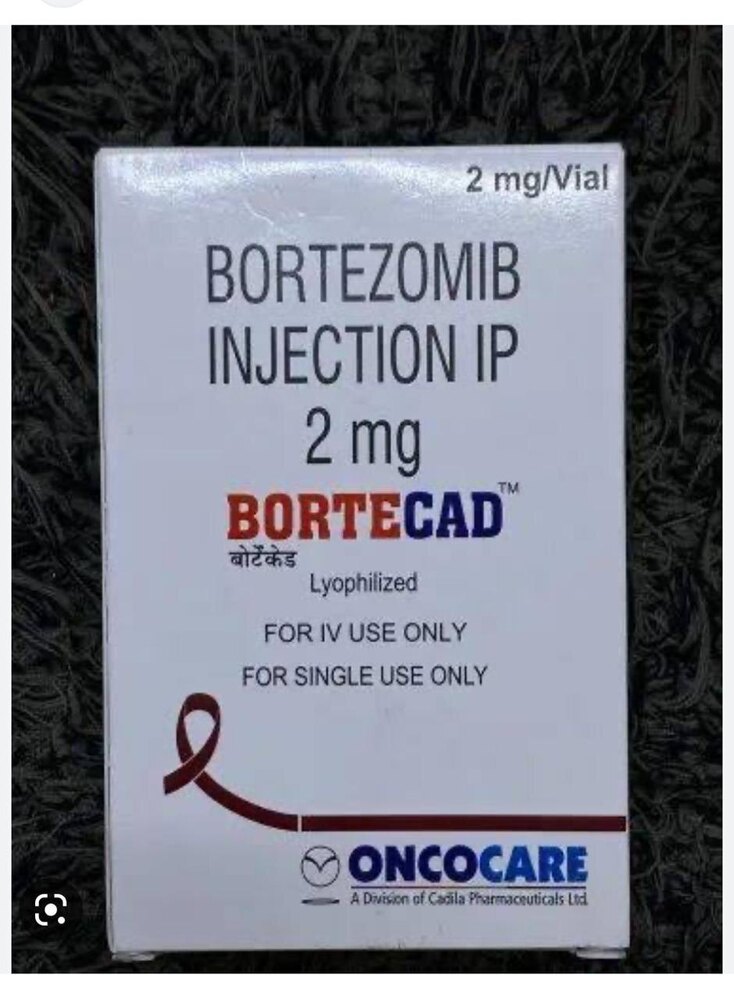

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
Ofloxacin & Ornidazole Tablets 200/500mg
Price 810 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Broad spectrum antibacterial and antiprotozoal activity
Function : Antimicrobial Therapy
Material : Other, Ofloxacin 200mg & Ornidazole 500mg
Storage Instructions : Store in a cool, dry & dark place, below 25C
Gastro-Resistant Omeprazole Capsules BP 10mg
Price 250 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Gastroresistant, entericcoated, delayed release
Function : Reduces gastric acid secretion
Material : Other, Gelatin (capsule shell), other excipients
Storage Instructions : Store below 25C, protect from moisture and light
Losartan Potassium Tablets USP 25mg
Price 250 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Reduces risk of stroke in patients with hypertension; comes in blister packaging
Function : Antihypertensive agent
Material : Other, Losartan Potassium
Storage Instructions : Store in a cool, dry place below 30C; protect from light and moisture
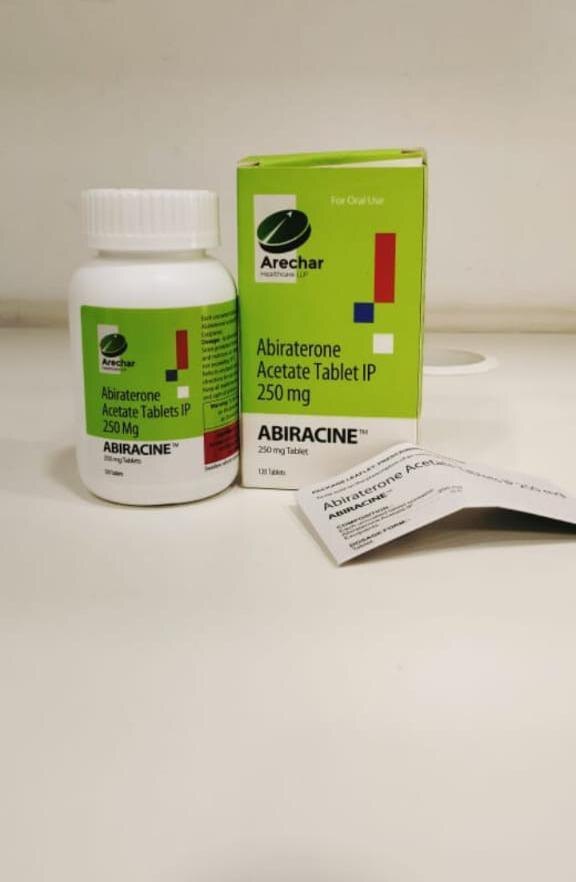
Abiraterone Acetate Tablets (ABIRACINE 250Mg)
Price 1500 INR / Pack
Minimum Order Quantity : 100 Packs
Features : 250 mg strength, easy to swallow, filmcoated tablet
Function : Antineoplastic agent, decreases androgen production
Material : Other, Pharmaceutical tablet
Storage Instructions : Store below 30C in a dry place, protect from light and moisture
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |