|
Borviz 2.5 Mg Injection
Borviz 2.5 Mg Injection Specification
- Measurement Range
- 2.5 mg/vial
- Function
- Antineoplastic agent (Bortezomib)
- Usage Type
- Injection
- Shelf Life
- 24 months from date of manufacture
- Accuracy
- Pharmaceutical grade precision
- Storage Instructions
- Store below 25C, protect from light and moisture, keep out of reach of children
- Frequency
- Dose as directed by physician
- Instruments Type
- Medicine vial
- Features
- Easy to reconstitute, Single-use vial, Anti-cancer agent, Prescription only
- Equipment Type
- Pharmaceutical Injection
- Material
- Sterile solution
- Condition
- New
- Technology
- Lyophilized Powder
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Parenteral (Intravenous or Subcutaneous)
- Use
- Multiple myeloma and mantle cell lymphoma treatment
- Dimension (L*W*H)
- Vial size standard (as per manufacturer packaging)
- Weight
- 2.5 mg active ingredient per vial
- Color
- White to off-white powder
- Prescription Status
- Prescription only
- Active Ingredient
- Bortezomib
- Box Contents
- 1 vial of lyophilized powder for injection
- Marketed By
- Intas Pharmaceuticals Ltd.
- Route of Administration
- IV or SC as recommended
- Caution
- Cytotoxic; handle with care
- Indication
- Treatment of multiple myeloma and mantle cell lymphoma
- Reconstitution Solvent
- As per package insert (typically 0.9% saline or sterile water)
- Drug Strength
- 2.5 mg per vial
- Pack Type
- Single vial
Borviz 2.5 Mg Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Borviz 2.5 Mg Injection
Borviz 2.5 mg Injection typically contains Bortezomib as the active ingredient. Bortezomib is a proteasome inhibitor used in the treatment of certain cancers, particularly multiple myeloma and mantle cell lymphoma.
Here are the key details about Borviz 2.5 mg Injection:
-
Active Ingredient: Bortezomib
-
Common Uses:
- Treatment of multiple myeloma, a cancer of the plasma cells in the bone marrow.
- Treatment of mantle cell lymphoma, a type of non-Hodgkin lymphoma.
-
Mechanism of Action:
- Bortezomib works by inhibiting the proteasome, an enzyme complex responsible for breaking down proteins within cells. By blocking the proteasome, it causes an accumulation of damaged proteins, leading to cell death, particularly in cancer cells that are rapidly dividing.
-
Dosage:
- The exact dosage and treatment schedule will be determined by the healthcare provider based on the patient's condition, age, and other factors. Typically, Borviz is administered through an intravenous (IV) injection or subcutaneously (under the skin).
-
Side Effects:
- Common side effects may include nausea, fatigue, diarrhea, fever, and low blood counts (such as low red or white blood cells).
- Serious side effects could include neuropathy (nerve damage), blood clotting problems, or heart-related issues.
-
Precautions:
- Close monitoring is required during treatment for potential side effects, especially related to blood counts and nerve function.
- It is essential to inform the healthcare provider if the patient has any history of heart problems, liver issues, or infections.
Advanced Cancer Treatment
Borviz 2.5 Mg Injection offers an effective solution for treating multiple myeloma and mantle cell lymphoma, leveraging the precision of Bortezomib. This medication inhibits the growth of cancer cells, providing significant benefit to patients when administered by a qualified healthcare professional.
Safe Handling & Storage
As a cytotoxic anti-cancer agent, Borviz must be handled with care. Store the vial below 25C and protect it from light and moisture. Always keep it out of reach of children, ensuring both safety and the preservation of the medication's efficacy.
User-Friendly Preparation
The vial contains a lyophilized powder that is easy to reconstitute, using 0.9% saline or sterile water as directed in the package insert. This enables timely preparation for clinicians and helps streamline the process for immediate use.
FAQ's of Borviz 2.5 Mg Injection:
Q: How should Borviz 2.5 Mg Injection be prepared before administration?
A: Borviz must be reconstituted according to the instructions in the package insert, typically using sterile water or 0.9% saline solution. The prepared solution is for single use only and should be administered promptly by a qualified medical professional.Q: What conditions is Borviz 2.5 Mg Injection used to treat?
A: Borviz 2.5 Mg Injection is indicated for the treatment of multiple myeloma and mantle cell lymphoma, providing an effective option for patients diagnosed with these forms of cancer.Q: When should Borviz 2.5 Mg Injection be administered?
A: The timing and frequency of administration should be determined by a physician, based on the patient's individual treatment plan and condition. Never self-administer; always follow the dosing schedule prescribed by your oncologist.Q: Where should Borviz 2.5 Mg Injection be stored?
A: Store the vial in a cool, dry place below 25C, protected from light and moisture. Keep the medication in its original packaging and away from children.Q: What is the process for administering Borviz 2.5 Mg Injection?
A: A healthcare provider will reconstitute the lyophilized powder and administer it either intravenously (IV) or subcutaneously (SC), following the recommended clinical guidelines and dosage instructions.Q: What precautions should be taken while handling Borviz 2.5 Mg Injection?
A: Borviz is a cytotoxic medication and should only be handled by trained professionals using appropriate protective equipment. Dispose of any unused materials according to standard cytotoxic drug procedures.Q: What are the benefits of using Borviz 2.5 Mg Injection in cancer treatment?
A: Borviz, with its active ingredient Bortezomib, specifically targets and disrupts cancer cell growth, improving outcomes and extending survival in patients with multiple myeloma and mantle cell lymphoma.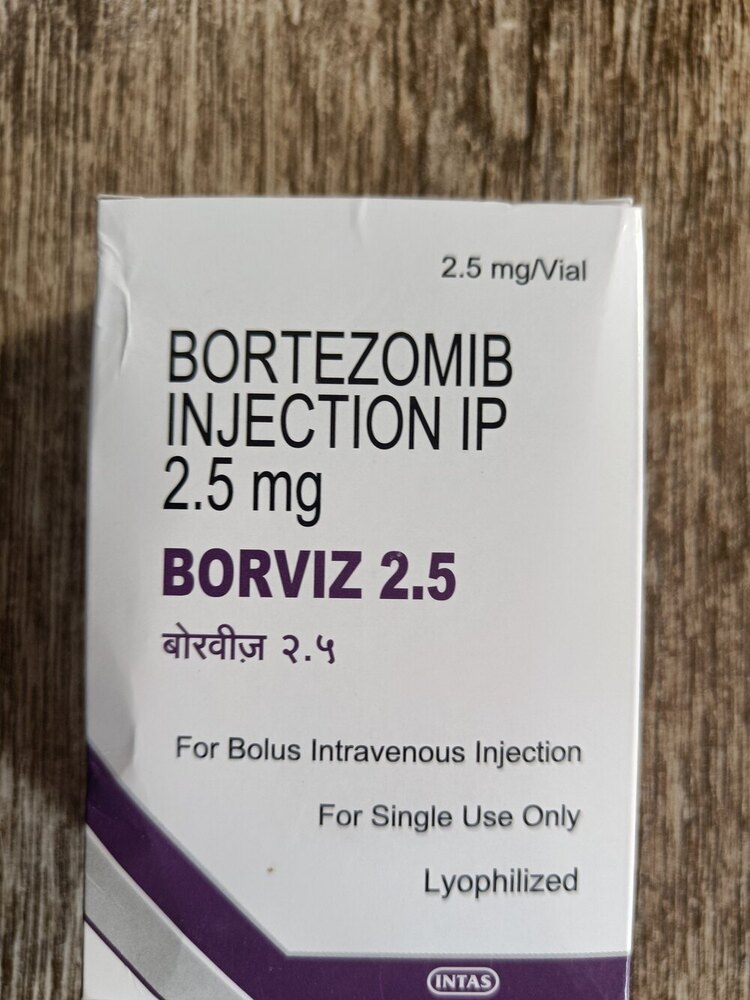

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
Coloplast Alterna 1 - Piece tranparent Bag 17455
Price 200 INR / Piece
Minimum Order Quantity : 100 Pieces
Features : Cuttofit opening, Secure outlet closure, Soft backing, Odorproof film
Condition : New
Use : Colostomy, Ileostomy, Urostomy
Shelf Life : 3 Years from manufacturing date
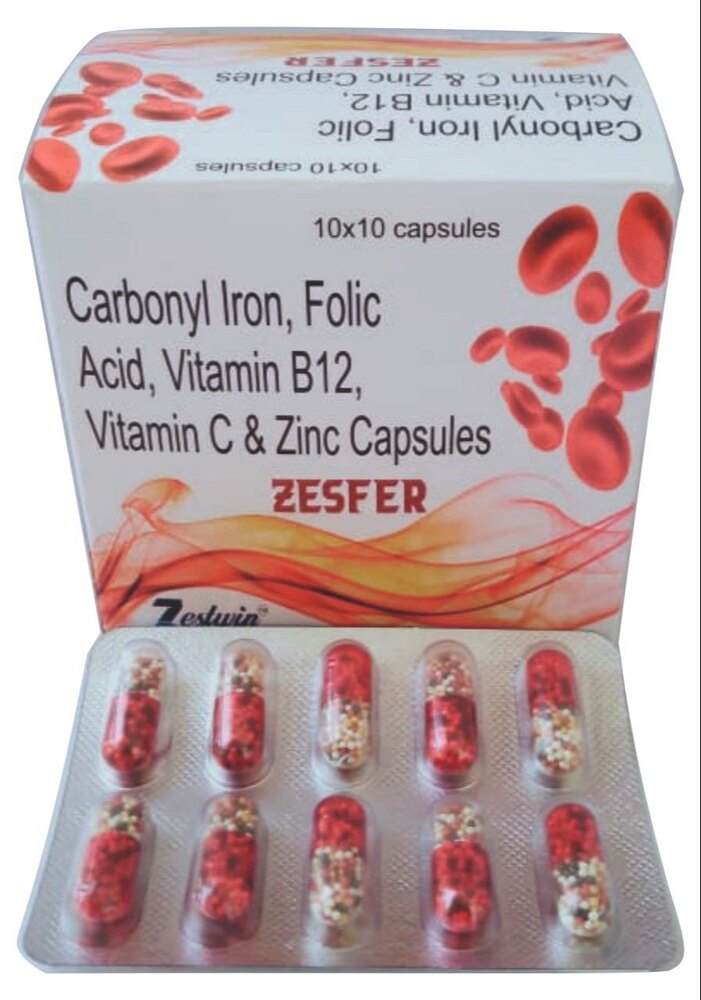
Iron, Folic Acid, Zinc & Vitamin B12 Capsule
Price 800 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Enriched with Iron, Folic Acid, Zinc & Vitamin B12; Promotes healthy blood; Supports immune system
Condition : New
Use : Nutritional Deficiency
Shelf Life : 24 Months
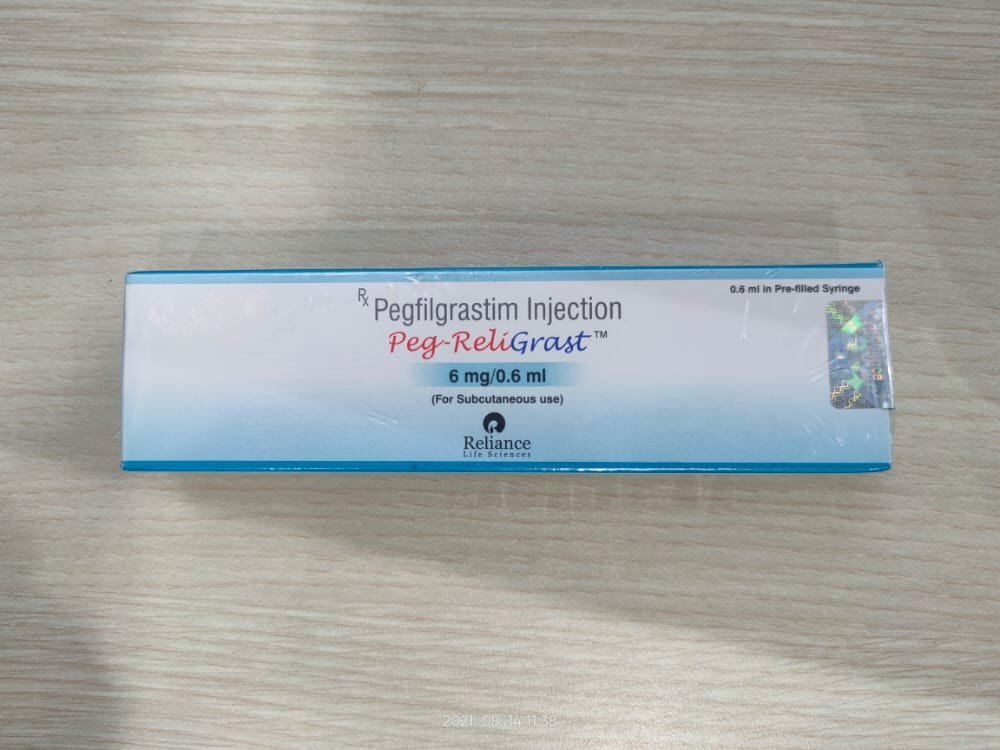
Pegreligrast Inj 6mg Pegfilgrastim
Price 6000 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Filgrastim pegylated for longer halflife, single use
Condition : New
Use : Medical, Oncology, Chemotherapy Induced Neutropenia
Shelf Life : 24 Months
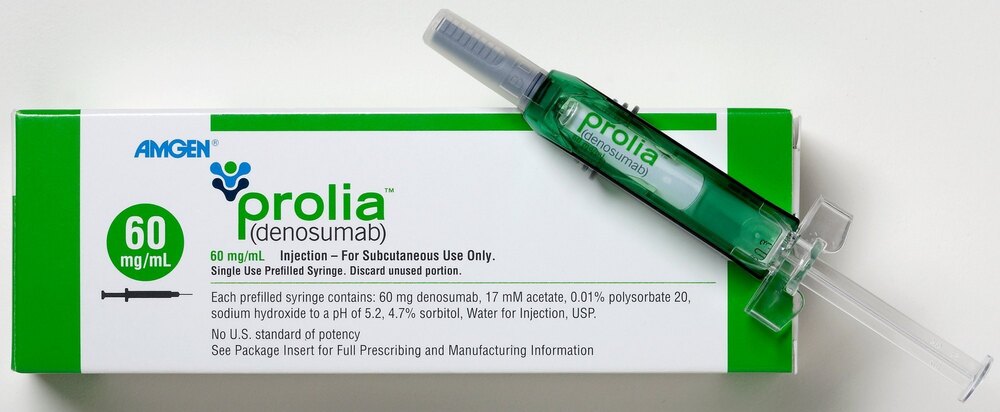
Prolia 60mg Injection
Price 10000 INR / Pack
Minimum Order Quantity : 100 Packs
Features : Readytouse prefilled syringe, singledose, easy administration
Condition : Other, New and sealed
Use : Treatment of osteoporosis and increased bone loss in adults
Shelf Life : 36 months from manufacturing date
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |