|
Bryxta 400 Mg Injection
Bryxta 400 Mg Injection Specification
- Usage Type
- Hospital, Clinical
- Shelf Life
- 24 Months
- Storage Instructions
- Store between 2C-8C, Do Not Freeze
- Function
- Anti-Cancer Agent, Monoclonal Antibody
- Features
- Ready to Use, Sterile, Preservative Free
- Instruments Type
- Therapeutic Injection
- Equipment Type
- Pharmaceutical Injection
- Material
- Sterile Solution
- Condition
- New
- Technology
- Biotechnology (Monoclonal Antibody)
- Portable
- Yes (in Single-Use Vial)
- Wall Mounted
- No
- Operating Type
- Parenteral (Intravenous Use)
- Use
- Cancer Treatment
- Color
- Transparent/Colorless
- Packing Type
- Vial
- Appearance
- Clear, colorless to pale yellow solution
- Administration Route
- Intravenous (IV) Infusion Only
- Indication
- Metastatic Colorectal Cancer, Non-Small Cell Lung Cancer, Ovarian Cancer, Other Solid Tumors
- Generic Name
- Bevacizumab
- Contraindications
- Hypersensitivity to Bevacizumab or any component of the formulation
- Dosage Form
- Injection
- Prescription/OTC
- Prescription Only
- Container Material
- Glass Vial
- Brand Name
- Bryxta
- Strength
- 400 mg
- Compatibility
- For use with compatible IV solutions
Bryxta 400 Mg Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Bryxta 400 Mg Injection
Bryxta 400 mg Injection contains Bruton's Tyrosine Kinase (BTK) Inhibitor known as Ibrutinib. It is a targeted therapy used in the treatment of certain types of cancer, particularly blood cancers.
Key Uses:
-
Chronic Lymphocytic Leukemia (CLL): Ibrutinib is commonly used to treat Chronic Lymphocytic Leukemia (CLL), a type of cancer that affects the white blood cells called B-cells.
-
Mantle Cell Lymphoma (MCL): It is also approved for the treatment of Mantle Cell Lymphoma, an aggressive form of non-Hodgkin lymphoma.
-
Waldenstrms Macroglobulinemia: Bryxta is used to treat Waldenstrms macroglobulinemia, a rare type of non-Hodgkin lymphoma that involves B-cells.
-
Other B-cell Malignancies: Bryxta may be used to treat other B-cell related cancers like Chronic Graft-versus-Host Disease (cGVHD) after stem cell transplants.
Mechanism of Action:
Ibrutinib works by inhibiting Bruton's Tyrosine Kinase (BTK), an enzyme that plays a crucial role in the signaling pathways of B-cells. BTK is involved in the activation of B-cells, which are critical for the growth and survival of many blood cancers. By inhibiting BTK, Ibrutinib blocks the signals that promote the growth and survival of these cancerous cells, leading to tumor shrinkage and cell death.
Common Side Effects:
- Fatigue or weakness.
- Diarrhea.
- Nausea.
- Bruising or bleeding more easily.
- Infections: Since Ibrutinib can lower the ability of the immune system to fight infections, there is an increased risk of infections.
- Rash or skin reactions.
Serious Side Effects:
- Heart Problems: Ibrutinib can cause arrhythmias or irregular heartbeats. Patients with existing heart problems should be monitored carefully.
- Bleeding: Ibrutinib can increase the risk of severe bleeding, including internal bleeding or major bruising.
- Infections: Due to its effect on the immune system, Ibrutinib may increase the risk of serious infections, such as pneumonia or fungal infections.
- Liver Toxicity: Liver problems (including elevated liver enzymes) can occur, requiring monitoring of liver function during treatment.
- High Blood Pressure: Ibrutinib can cause high blood pressure (hypertension), which should be managed during treatment.
Dosage:
The typical dose of Bryxta 400 mg will depend on the condition being treated and the patient's individual response. It is usually administered as a single oral dose, but the 400 mg dose is generally given once daily.
Administration:
Bryxta is administered via intravenous (IV) infusion. It is typically given in a clinical setting, with the administration taking place over a period of time, depending on the dose and the patient's needs.
Precautions:
- Heart and Blood Pressure: Patients should be monitored for heart issues or high blood pressure, as Ibrutinib can cause arrhythmias and elevated blood pressure.
- Infection Risk: Ibrutinib weakens the immune system, so precautions should be taken to prevent infections.
- Liver Function: Regular monitoring of liver function tests is necessary as liver toxicity can occur.
- Bleeding Risk: Patients should be monitored for signs of bleeding, such as unusual bruising, blood in the urine, or excessive bleeding during minor cuts or injuries.
- Pregnancy: Ibrutinib is contraindicated during pregnancy and breastfeeding due to potential harm to the fetus or infant. Women of childbearing age should use reliable contraception during treatment.
Summary:
Bryxta 400 mg Injection contains Ibrutinib, a Bruton's Tyrosine Kinase (BTK) inhibitor, used to treat Chronic Lymphocytic Leukemia (CLL), Mantle Cell Lymphoma (MCL), and other B-cell related cancers. It works by blocking a key enzyme in B-cells, which slows or stops the growth of cancerous cells. Side effects include fatigue, diarrhea, and an increased risk of infections, bleeding, and heart problems. Regular monitoring of liver function, blood pressure, and signs of bleeding is essential during treatment. Ibrutinib should not be used in pregnant women and requires careful management to prevent serious side effects.
Advanced Cancer Treatment with Biotechnology
Bryxta 400 Mg Injection offers a modern biotechnology solution in cancer therapeutics. Using Bevacizumab, it targets tumor growth in patients with metastatic colorectal cancer, non-small cell lung cancer, and other solid tumors. Its monoclonal antibody action helps inhibit tumor blood supply, enhancing treatment outcomes and making it an integral part of contemporary oncology care.
Convenience and Safety in Hospital Settings
Designed for hospital and clinical environments, Bryxta 400 Mg Injection is provided in a ready-to-use, sterile vial. Its transparent, colorless solution ensures visual clarity for professional administration. The product's packaging and storage requirements promote optimal therapeutic effectiveness and patient safety throughout its shelf life.
FAQ's of Bryxta 400 Mg Injection:
Q: How is Bryxta 400 Mg Injection administered?
A: Bryxta 400 Mg Injection is given as an intravenous (IV) infusion by a trained healthcare professional in a hospital or clinical setting. The dose and duration depend on the specific type of cancer and patient condition.Q: What types of cancer can Bryxta 400 Mg Injection treat?
A: Bryxta is used for the management of metastatic colorectal cancer, non-small cell lung cancer, ovarian cancer, and other solid tumors as determined by your oncologist.Q: When should Bryxta 400 Mg Injection be used?
A: Bryxta is prescribed by physicians when a diagnosis indicates it is appropriate for cancer treatment. The injection is usually used as part of a combination regimen, based on the patient's clinical needs.Q: Where should Bryxta 400 Mg Injection be stored?
A: This injection should be kept in a refrigerator at 2C-8C. Do not freeze the product, as improper storage may compromise its effectiveness and safety.Q: What is the recommended process for using Bryxta 400 Mg Injection?
A: A healthcare provider dilutes Bryxta in a compatible IV solution and administers it slowly through an intravenous infusion. It must never be injected directly or administered without dilution, and only single-use vials are used per session.Q: What are the benefits of Bryxta 400 Mg Injection?
A: Bryxta may enhance the effectiveness of cancer therapy by inhibiting blood supply to tumors, potentially slowing cancer progression. Its biotechnology-based monoclonal antibody approach represents an advanced method in anti-cancer treatment.Q: Who should not receive Bryxta 400 Mg Injection?
A: Patients with known hypersensitivity to Bevacizumab or any formulation component must not use Bryxta. Always inform healthcare providers about allergies before beginning treatment.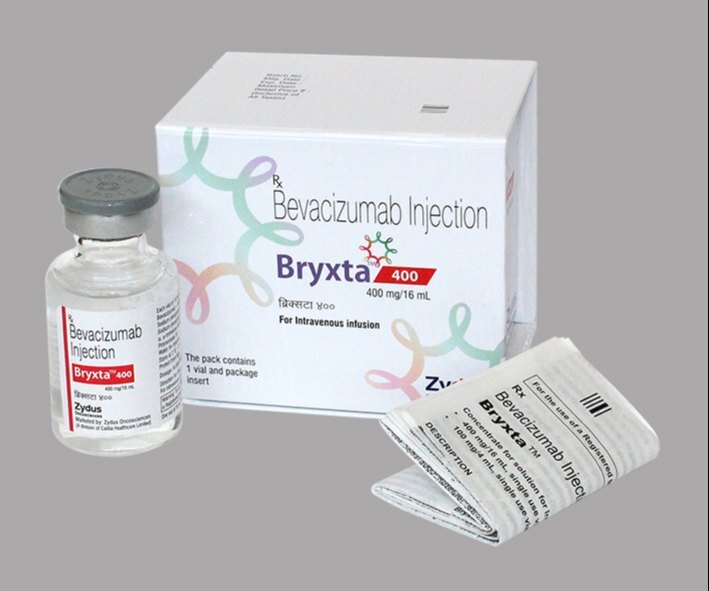

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
Coloplast Sensura Convex Light 1 Piece drainable bag 15205
Price 250 INR / Piece
Minimum Order Quantity : 100 Pieces
Material : Other, Soft, odorproof plastic with woven textile backing
Equipment Type : Ostomy Bag
Usage Type : SingleUse
Portable : Yes
Magic Touch Sirolimus Coated Balloon Catheter
Price 6400 INR / Pack
Minimum Order Quantity : 100 Packs
Material : Other, Polymer/Polyamide balloon with Drug Coating
Equipment Type : Sirolimus Coated Balloon Catheter
Usage Type : Hospital/Clinical
Portable : Yes
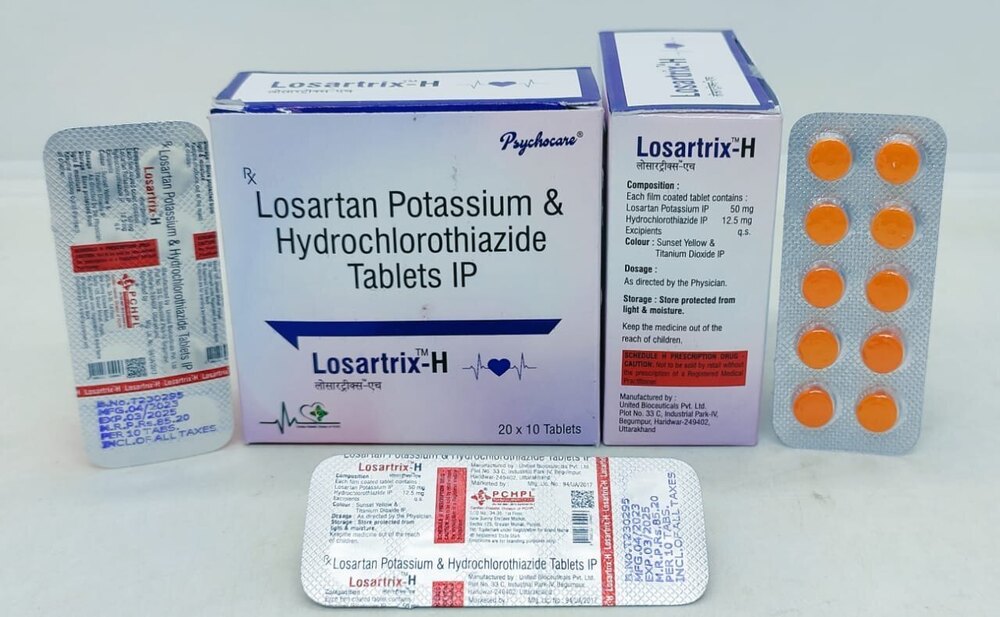
Losartan Potassium and Hydrochlorothiazide Tablets USP 50/12.5mg
Price 85 INR / Pack
Minimum Order Quantity : 100 Packs
Material : Other, Losartan Potassium and Hydrochlorothiazide USP
Equipment Type : Pharmaceutical Tablet
Usage Type : Prescription Only (Rx)
Portable : Yes
Amino Acids & Multivitamin Syrup
Price 150 INR / Pack
Minimum Order Quantity : 100 Packs
Material : Other, Liquid Syrup
Equipment Type : Amino Acids & Multivitamin Syrup
Usage Type : Internal
Portable : Yes
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |