|
Eleftha 150mg Injection
Eleftha 150mg Injection Specification
- Instruments Type
- Injectable vial
- Function
- Stimulates erythropoiesis
- Shelf Life
- 24 Months
- Accuracy
- Pharmaceutical grade
- Frequency
- As prescribed by physician
- Usage Type
- Prescription medicine
- Storage Instructions
- Store at 2C to 8C, do not freeze, keep away from light
- Features
- Pre-filled vial, single use, preservative-free
- Measurement Range
- 150mg per vial
- Equipment Type
- Pharmaceutical Injection
- Material
- Liquid Solution in Vial
- Condition
- New
- Technology
- Recombinant Human Erythropoietin Alfa
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Parenteral
- Use
- Used in the treatment of Anemia associated with chronic renal failure and cancer chemotherapy
- Dimension (L*W*H)
- Per unit packaging size (vial)
- Weight
- Lightweight (single dose vial)
- Color
- Clear, colorless liquid
- Therapeutic Class
- Hematopoietic agent
- Dosage Form
- Injection
- Packing Type
- Single vial
- Marketed By
- Intas Pharmaceuticals
- Prescription Required
- Yes
- Brand Name
- Eleftha 150mg Injection
- Expiry Date
- As per printed label
- Route of Administration
- Intravenous or Subcutaneous
- Generic Name
- Erythropoietin Alfa Injection
- Indication
- Anemia due to chronic kidney disease or chemotherapy
Eleftha 150mg Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- FOB Port
- AHMEDABAD
- Payment Terms
- Paypal, Cash Advance (CA), Cash in Advance (CID), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Delivery Time
- 3 Days
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Box
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Eleftha 150mg Injection
Eleftha 150 mg Injection contains Efalizumab, a monoclonal antibody primarily used in the treatment of moderate to severe psoriasis, particularly psoriatic arthritis or chronic plaque psoriasis. Efalizumab works by modifying the immune response, targeting specific proteins involved in the immune system's activity.
Indications
Eleftha (Efalizumab) is used for:
-
Psoriasis: It is primarily indicated for the treatment of moderate to severe chronic plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy. Psoriasis is an autoimmune condition that causes the rapid turnover of skin cells, resulting in scaly, inflamed patches on the skin.
-
Psoriatic Arthritis: In some cases, it may also be used to treat psoriatic arthritis, where the skin and joints are affected by inflammation due to the immune system attacking healthy tissues.
Mechanism of Action
Efalizumab is a monoclonal antibody that works by binding to CD11a, a subunit of the LFA-1 (Lymphocyte Function-Associated Antigen-1) protein, found on the surface of white blood cells. LFA-1 is involved in the interaction between immune cells (T-cells) and other cells, including those in the skin, and plays a key role in the inflammatory response.
By blocking the activity of LFA-1, Efalizumab prevents T-cells from migrating to the skin and causing the inflammatory response seen in psoriasis. This reduces inflammation and the rapid turnover of skin cells, helping to control the skin symptoms of psoriasis.
Dosage and Administration
-
Typical Dosage:
- Initial Dose: The usual dose for Eleftha (Efalizumab) is 1 mg/kg, administered as a subcutaneous injection (under the skin). This is typically given once a week.
- Maintenance Dose: After the initial dose, the maintenance dose is 1 mg/kg once a week. The injection is usually administered in a clinical setting, but it may also be given at home under medical supervision.
-
Administration:
- Subcutaneous Injection: Efalizumab is injected subcutaneously, which means it is injected into the fatty tissue under the skin, typically in the thigh, abdomen, or upper arm.
- Injection Sites: The injection site should be rotated to avoid irritation.
-
Renal or Hepatic Impairment: There are no specific dose adjustments required for patients with renal or hepatic impairment, but regular monitoring of liver and kidney function is recommended during treatment.
Side Effects
Common side effects of Eleftha (Efalizumab) include:
- Headache
- Fever
- Nausea
- Fatigue
- Injection site reactions: Such as redness, pain, or swelling at the injection site.
- Rashes: Some patients may experience rashes that are part of the drug's effect or related to the underlying condition.
Serious side effects (though less common) may include:
- Infections: Efalizumab works by suppressing parts of the immune system, which may increase the risk of infections, including upper respiratory infections, urinary tract infections, or even more serious infections like tuberculosis.
- Progressive Multifocal Leukoencephalopathy (PML): This is a rare but serious brain infection that can occur when the immune system is significantly suppressed. Patients should be monitored for any neurological symptoms.
- Hepatitis B Reactivation: In rare cases, Efalizumab can lead to the reactivation of hepatitis B infection. Liver function tests are essential during treatment.
- Allergic Reactions: Anaphylaxis (severe allergic reactions) is a rare side effect that requires immediate medical attention.
Precautions and Warnings
- Infections: Since Efalizumab suppresses the immune system, it can increase the susceptibility to infections. Patients should be screened for tuberculosis and other infections before starting treatment.
- Neurological Effects: Neurological symptoms such as confusion, changes in vision, or difficulty speaking should be reported to a healthcare provider immediately as they could indicate PML or other serious complications.
- Hepatitis B: Patients with a history of hepatitis B should be carefully monitored, as the drug can trigger a reactivation of the virus in some cases.
- Vaccinations: Patients should avoid live vaccines while on Efalizumab due to the suppression of the immune system. Non-live vaccines may be recommended before treatment begins.
- Pregnancy: Efalizumab is categorized as a Category C drug, meaning its safety during pregnancy has not been fully established. It should only be used during pregnancy if the potential benefits outweigh the risks.
- Breastfeeding: It is not known if Efalizumab passes into breast milk. It should be used cautiously in breastfeeding mothers, and alternative treatments may be considered.
Important Considerations
- Regular Monitoring: Because Eleftha (Efalizumab) can affect the immune system, regular blood tests and monitoring for signs of infection are essential during treatment. This includes monitoring for signs of PML, liver issues, and infections.
- Adherence to Treatment: Patients are encouraged to adhere strictly to the prescribed weekly dosing schedule for the best results.
- Injection Technique: It is important that injections are performed correctly to avoid complications like infection at the injection site. Training may be provided for patients who will administer the injections at home.
Conclusion
Eleftha 150 mg Injection (Efalizumab) is a monoclonal antibody used to treat moderate to severe chronic plaque psoriasis and possibly psoriatic arthritis by modifying the immune response. It works by blocking T-cell migration to the skin, reducing inflammation and cell turnover in psoriasis. Although effective, Efalizumab can suppress the immune system, increasing the risk of infections and requiring careful monitoring throughout treatment.
Targeted Anemia Management
Eleftha 150mg Injection is formulated to specifically target anemia stemming from chronic kidney disease and chemotherapy. Its active ingredient, Erythropoietin Alfa, effectively boosts red blood cell production, leading to improved energy levels and overall well-being for patients who suffer from anemia due to these medical conditions.
Easy-to-Administer Injectable Vial
Packed in a pre-filled, single-use vial, Eleftha 150mg is designed for flexible administration by healthcare professionals. It can be given intravenously or subcutaneously, ensuring that the method best suited to the patient's requirements is employed. Its preservative-free formulation adds to its safety and suitability.
Safe Storage and Reliable Shelf Life
To maintain its efficacy, Eleftha 150mg Injection should be stored at 2C to 8C, never frozen, and protected from light. Each vial maintains pharmaceutical-grade accuracy for up to 24 months from the date of manufacture, ensuring both potency and patient safety over its shelf life.
FAQ's of Eleftha 150mg Injection:
Q: How is Eleftha 150mg Injection administered?
A: Eleftha 150mg Injection is intended for parenteral use and can be administered either intravenously or subcutaneously by a healthcare professional, as directed by a physician.Q: What conditions does Eleftha 150mg Injection treat?
A: This injection is indicated for the management of anemia associated with chronic kidney disease or resulting from cancer chemotherapy.Q: When should Eleftha 150mg Injection be used?
A: Eleftha 150mg Injection should be used strictly as prescribed by your physician, who will determine the appropriate time and frequency based on your medical condition and response to therapy.Q: Where should Eleftha 150mg Injection be stored?
A: Store the vial in a refrigerator at 2C to 8C. Do not freeze, and ensure the vial is kept away from direct light to preserve its efficacy.Q: What are the benefits of using Eleftha 150mg Injection?
A: The primary benefit is increased red blood cell production, which helps alleviate symptoms of anemia such as fatigue and weakness, thereby improving oxygen delivery and quality of life.Q: What is the process for using Eleftha 150mg Injection?
A: A healthcare professional will prepare and administer the injection from the pre-filled single-use vial, ensuring the correct dose is given via the appropriate route as per your medical requirements.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Skin Care Products Category
Coloplast Brava Adhesive Remover Wipes 12011
Price 35000.0 INR / Pack
Minimum Order Quantity : 100 Packs
Equipment Type : Adhesive Remover Wipes
Wall Mounted : No
Function : Removes stoma appliance adhesives gently and effectively
Color : White wipe, blue/white sachet
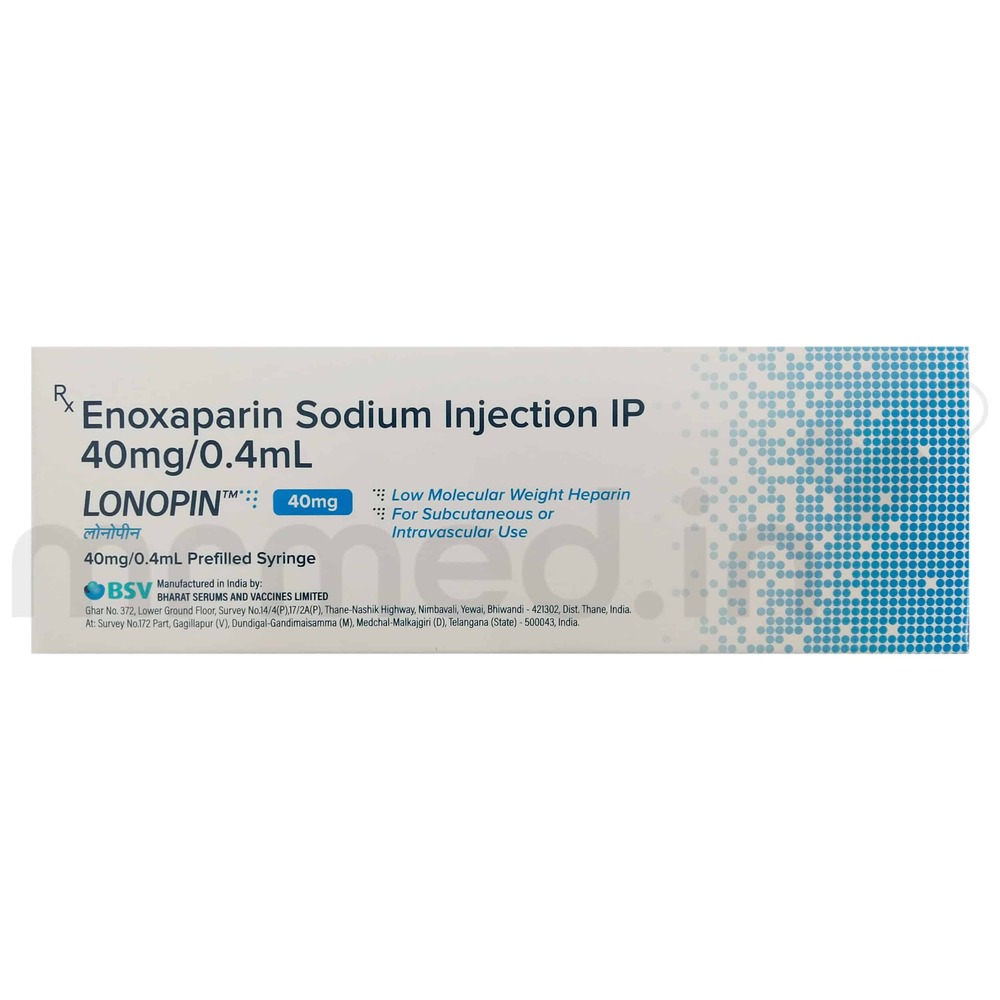
Lonopin 40 Mg Injection
Price 180 INR / Pack
Minimum Order Quantity : 100 Packs
Equipment Type : Injection
Wall Mounted : No
Function : Prevention of deep vein thrombosis (DVT), anticoagulant
Color : Transparent with yellow label
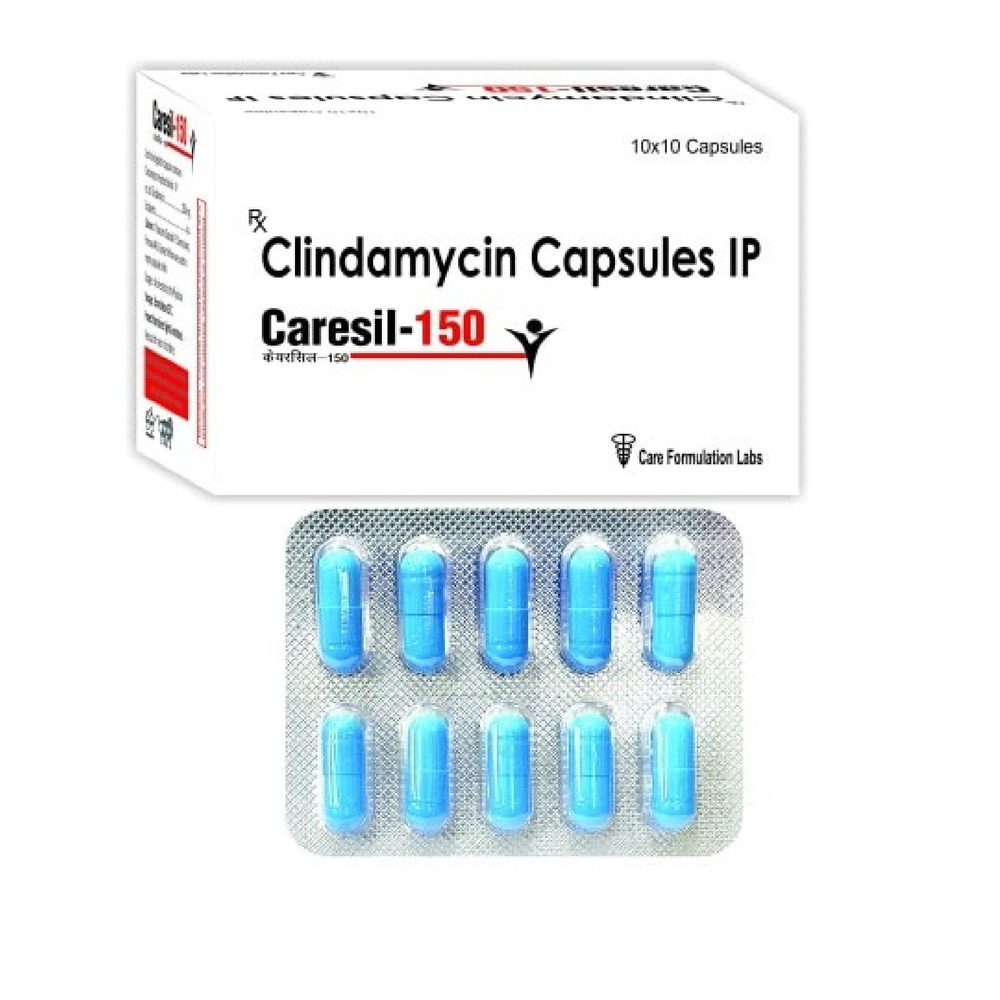
Clindamycin Capsules BP 150mg
Price 150 INR / Pack
Minimum Order Quantity : 100 Packs
Equipment Type : Pharmaceutical Capsule
Wall Mounted : No
Function : Antibiotic
Color : White
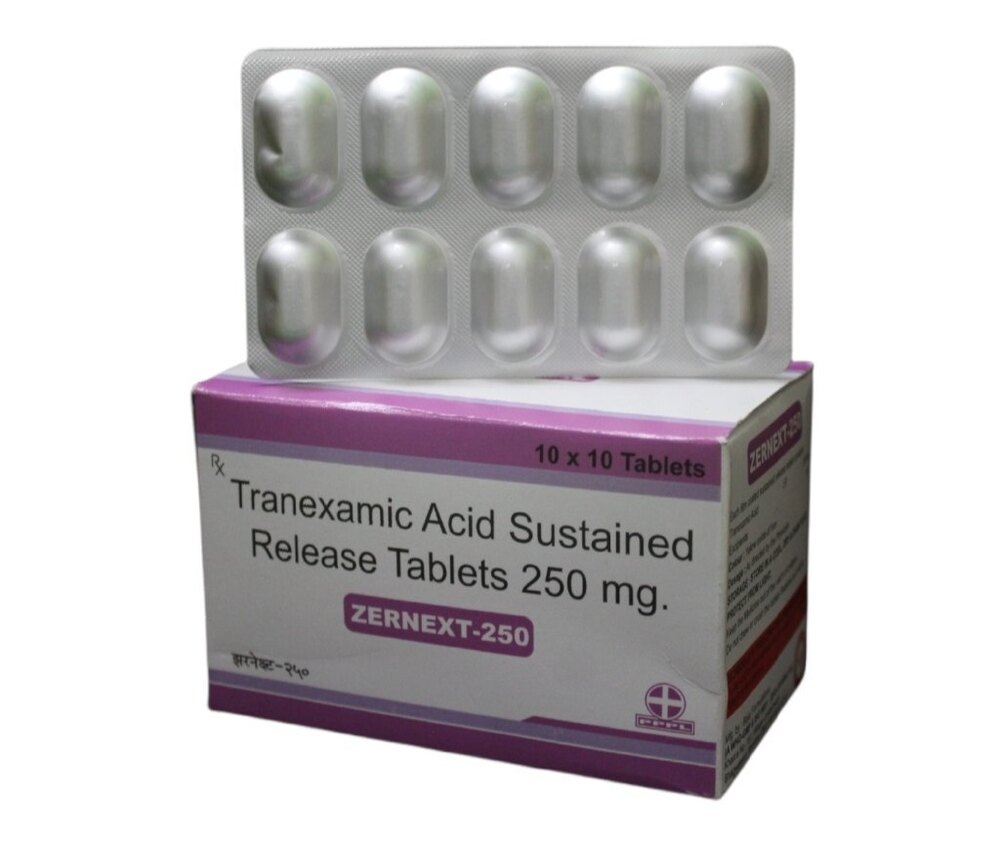
Tranexamic Acid Tablets BP 250mg
Price 160 INR / Pack
Minimum Order Quantity : 100 Packs
Equipment Type : Pharmaceutical Tablet
Wall Mounted : No
Function : Antifibrinolytic agent to reduce bleeding
Color : White
 |
TABNCAP HEALTHCARE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |